|

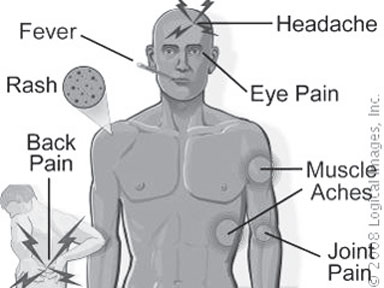
Strategy to disarm the virus: New hope for a universal dengue
vaccine
A new strategy that cripples the ability of the dengue virus to
escape the host immune system has been discovered by A STAR's Singapore
Immunology Network (SIgN). This breakthrough strategy opens a door of
hope to what may become the world's first universal dengue vaccine
candidate that can give full protection from all four serotypes of the
dreadful virus.
 Early studies have shown that a sufficiently weakened virus that is
still strong enough to generate protective immune response offers the
best hope for an effective vaccine. However, over the years of vaccine
development, scientists have learnt that the path to finding a virus of
appropriate strength is fraught with challenges. This hurdle is
compounded by the complexity of the dengue virus. Even though there are
only four different serotypes, the fairly high rates of mutation means
the virus evolve constantly, and this contributes to the great diversity
of the dengue viruses circulating globally. Early studies have shown that a sufficiently weakened virus that is
still strong enough to generate protective immune response offers the
best hope for an effective vaccine. However, over the years of vaccine
development, scientists have learnt that the path to finding a virus of
appropriate strength is fraught with challenges. This hurdle is
compounded by the complexity of the dengue virus. Even though there are
only four different serotypes, the fairly high rates of mutation means
the virus evolve constantly, and this contributes to the great diversity
of the dengue viruses circulating globally.
In some cases, the immune response developed following infection by
one of the four dengue viruses appears to increase the risk of severe
dengue when the same individual is infected with any of the remaining
three viruses.
With nearly half the world's population at risk of dengue infection
and an estimated 400 million people getting infected each year[2], the
need for a safe and long-lasting vaccine has never been greater.
The new strategy uncovered in this study overcomes the prevailing
challenges of vaccine development by tackling the virus’ ability to
‘hide’ from the host immune system. Dengue virus requires the enzyme
called Mtase (also known as 2'-O-methyltransferase) to chemically modify
its genetic material to escape detection. In this study, the researchers
discovered that by introducing a genetic mutation to deactivate the
MTase enzyme of the virus, initial cells infected by the weakened MTase
mutant virus is immediately recognised as foreign. As a result, the
desired outcome of a strong protective immune response is triggered yet
at the same time the mutant virus hardly has a chance to spread in the
host.
Animal models immunised with the weakened MTase mutant virus were
fully protected from a challenge with the normal dengue virus. The
researchers went on to demonstrate that the MTase mutant dengue virus
cannot infect Aedes mosquitoes. This means that the mutated virus is
unable to replicate in the mosquito, and will not be able to spread
through mosquitoes into our natural environment. Taken together, the
results confirmed that Mtase mutant dengue virus is potentially a safe
vaccine approach for developing a universal dengue vaccine that protects
from all four serotypes.
The team leader, Dr Katja Fink from SIgN said, “There is still no
clinically approved vaccine or specific treatment available for dengue,
so we are very encouraged by the positive results with this novel
vaccine strategy.
Our next step will be to work on a vaccine formulation that will
confer full protection from all four serotypes with a single injection.
If this proves to be safe in humans, it can be a major breakthrough for
the dengue vaccine field.”
Associate Prof Leo Yee Sin, who heads the Singapore STOP Dengue
Translational and Clinical Research (TCR) Program said, “We are into the
seventh decade of dengue vaccine development, this indeed is an exciting
breakthrough that brings us a step closer to an effective vaccine.”
Associate Professor Laurent Rénia said, “Dengue is a major public
health problem in many of the tropical countries. We are very delighted
that our collaborative efforts with colleagues in Singapore and China
have made a promising step towards a cost-effective and safe dengue
vaccine to combat the growing threat of dengue worldwide.”
The Singapore Immunology Network (SIgN), officially inaugurated on
January 15, 2008, is a research consortium under the Agency for Science,
Technology and Research (A*STAR)'s Biomedical Research Council.
The mandate of SIgN is to advance human immunology research and
participate in international efforts to combat major health problems.
Since its launch, SIgN has grown rapidly and currently includes 250
scientists from 26 different countries around the world working under 28
renowned principal investigators.
Through this, SIgN aims at building a strong platform in basic human
immunology research for better translation of research findings into
clinical applications.
SIgN also sets out to establish productive links with local and
international institutions, and encourage the exchange of ideas and
expertise between academic, industrial and clinical partners and thus
contribute to a vibrant research environment in Singapore.
MNT
A well-connected core brain network helps humans to adapt
One thing that sets humans apart from other animals is our ability to
intelligently and rapidly adapt to a wide variety of new challenges -
using skills learned in much different contexts to inform and guide the
handling of any new task at hand.
Now, research from Washington University in St. Louis offers new and
compelling evidence that a well-connected core brain network based in
the lateral prefrontal cortex and the posterior parietal cortex - parts
of the brain most changed evolutionarily since our common ancestor with
chimpanzees - contains “flexible hubs” that coordinate the brain's
responses to novel cognitive challenges.
 Acting as a central switching station for cognitive processing, this
fronto-parietal brain network funnels incoming task instructions to
those brain regions most adept at handling the cognitive task at hand,
coordinating the transfer of information among processing brain regions
to facilitate the rapid learning of new skills, the study finds. Acting as a central switching station for cognitive processing, this
fronto-parietal brain network funnels incoming task instructions to
those brain regions most adept at handling the cognitive task at hand,
coordinating the transfer of information among processing brain regions
to facilitate the rapid learning of new skills, the study finds.
“Flexible hubs are brain regions that coordinate activity throughout
the brain to implement tasks - like a large Internet traffic router,”
suggests Michael Cole, lead author of the study.
“Like an Internet router, flexible hubs shift which networks they
communicate with based on instructions for the task at hand and can do
so even for tasks never performed before,” he adds.
Decades of brain research has built a consensus understanding of the
brain as an interconnected network of as many as 300 distinct regional
brain structures, each with its own specialised cognitive functions.
Binding these processing areas together is a web of about a dozen
major networks, each serving as the brain's means for implementing
distinct task functions - i.e. auditory, visual, tactile, memory,
attention and motor processes. It was already known that fronto-parietal
brain regions form a network that is most active during novel or
non-routine tasks, but it was unknown how this network's activity might
help implement tasks.
This study proposes and provides strong evidence for a “flexible hub”
theory of brain function in which the fronto-parietal network is
composed of flexible hubs that help to organise and coordinate
processing among the other specialised networks.
This study provides strong support for the flexible hub theory in two
key areas.
First, the study yielded new evidence that when novel tasks are
processed flexible hubs within the fronto-parietal network make
multiple, rapidly shifting connections with specialised processing areas
scattered throughout the brain.
Second, by closely analysing activity patterns as the flexible hubs
connect with various brain regions during the processing of specific
tasks, researchers determined that these connection patterns include
telltale characteristics that can be decoded and used to identify which
specific task is being implemented by the brain.
These unique patterns of connection - like the distinct strand
patterns of a spider web - appear to be the brain's mechanism for the
coding and transfer of specific processing skills, the study suggests.
By tracking where and when these unique connection patterns occur in
the brain, researchers were able to document flexible hubs’ role in
shifting previously learned and practised problem-solving skills and
protocols to novel task performance. Known as compositional coding, the
process allows skills learned in one context to be re-packaged and
re-used in other applications, thus shortening the learning curve for
novel tasks.
What's more, by tracking the testing performance of individual study
participants, the team demonstrated that the transfer of these
processing skills helped participants speed their mastery of novel
tasks, essentially using previously practised processing tricks to get
up to speed much more quickly for similar challenges in a novel setting.
“The flexible hub theory suggests this is possible because flexible hubs
build up a repertoire of task component connectivity patterns that are
highly practiced and can be reused in novel combinations in situations
requiring high adaptivity,” Cole explains.
“It's as if a conductor practised short sound sequences with each
section of an orchestra separately, then on the day of the performance
began gesturing to some sections to play back what they learned,
creating a new song that has never been played or heard before.”
By improving our understanding of cognitive processes behind the
brain's handling of novel situations, the flexible hub theory may one
day help us improve the way we respond to the challenges of everyday
life, such as when learning to use new technology, Cole suggests.
“Additionally, there is evidence building that flexible hubs in the
fronto-parietal network are compromised for individuals suffering from a
variety of mental disorders, reducing the ability to effectively
self-regulate and thereforeexacerbating symptoms,” he says.
Future research may provide the means to enhance flexible hubs in
ways that would allow people to increase self-regulation and reduce
symptoms in a variety of mental disorders, such as depression,
schizophrenia and obsessive-compulsive disorder.
- MNT
Cancer cells change while moving throughout body
For the majority of cancer patients, it's not the primary tumour;
that is deadly, but the spread or “metastasis” of cancer cells from the
primary tumour to secondary locations throughout the body that is the
problem. That's why a major focus of contemporary cancer research is how
to stop or fight metastasis.
Previous lab studies suggest that metastasising cancer cells undergo
a major molecular change when they leave the primary tumour - a process
called epithelial-to-mesenchymal transition (EMT). As the cells travel
from one site to another, they pick up new characteristics. More
importantly, they develop a resistance to chemotherapy that is effective
on the primary tumour. But confirmation of the EMT process has only
taken place in test tubes or in animals.
 In a study, in the Journal of Ovarian Research, Georgia Tech
scientists have direct evidence that EMT takes place in humans, at least
in ovarian cancer patients. The findings suggest that doctors should
treat patients with a combination of drugs: those that kill cancer cells
in primary tumours and drugs that target the unique characteristics of
cancer cells spreading through the body. In a study, in the Journal of Ovarian Research, Georgia Tech
scientists have direct evidence that EMT takes place in humans, at least
in ovarian cancer patients. The findings suggest that doctors should
treat patients with a combination of drugs: those that kill cancer cells
in primary tumours and drugs that target the unique characteristics of
cancer cells spreading through the body.
The researchers looked at matching ovarian and abdominal cancerous
tissues in seven patients. Pathologically, the cells looked exactly the
same, implying that they simply fell off the primary tumour and spread
to the secondary site with no changes. But on the molecular level, the
cells were very different. Those in the metastatic site displayed
genetic signatures consistent with EMT. The scientists didn't see the
process take place, but they know it happened.
“It's like noticing that a piece of cake has gone missing from your
kitchen and you turn to see your daughter with chocolate on her face,”
said John McDonald, lead investigator on the project. “You didn't see
her eat the cake, but the evidence is overwhelming. The gene expression
patterns of the metastatic cancers displayed gene expression profiles
that unambiguously identified them as having gone through EMT.”
The EMT process is an essential component of embryonic development
and allows for reduced cell adhesiveness and increased cell movement.
According to Benedict Benigno, director of gynaecological oncology at
Atlanta's Northside Hospital, “These results clearly indicate that
metastasising ovarian cancer cells are very different from those
comprising the primary tumour and will likely require new types of
chemotherapy if we are going to improve the outcome of these patients.”
Ovarian cancer is the most malignant of all gynaecological cancers
and responsible for more than 14,000 deaths annually in the United
States alone. It often reveals no early symptoms and isn't typically
diagnosed until after it spreads.
“Our team is hopeful that, because of the new findings, the
substantial body of knowledge that has already been acquired on how to
block EMT and reduce metastasis in experimental models may now begin to
be applied to humans,” said Loukia Lili, co-author of the study.
- MNT
Seeking a clinical test for breast cancer
An international scientific collaborative led by the Harvard Stem
Cell Institute's Kornelia Polyak, MD, PhD, has discovered why women who
give birth in their early twenties are less likely to eventually develop
breast cancer than women who don't, triggering a search for a way to
confer this protective state on all women.
 The researchers now are in the process of testing p27, a mammary
gland progenitor marker, in the tissue of thousands of women collected
over a 20-year period - women whose histories have been followed
extremely closely - to see if it is an accurate breast cancer predictor
in a large population of women. If the hypothesis is confirmed, likely
within a few months, Polyak says the commercial development of a
clinical test for breast cancer risk would follow. The researchers now are in the process of testing p27, a mammary
gland progenitor marker, in the tissue of thousands of women collected
over a 20-year period - women whose histories have been followed
extremely closely - to see if it is an accurate breast cancer predictor
in a large population of women. If the hypothesis is confirmed, likely
within a few months, Polyak says the commercial development of a
clinical test for breast cancer risk would follow.
In a paper in Cell Stem Cell, the researchers describe how a
full-term pregnancy in a woman's early twenties reduces the relative
number and proliferative capacity of mammary gland progenitors - cells
that have the ability to divide into milk-producing cells – making them
less likely to acquire mutations that lead to cancer.
By comparing numerous breast tissue samples, the scientists found
that women at high risk for breast cancer, such as those who inherit a
mutated BRCA1 or BRCA2 gene, have higher-than-average numbers of mammary
gland progenitors. In general, women who carried a child to full term
had the lowest populations of mammary gland progenitors, even when
compared to cancer-free women who had never been pregnant. In addition,
in woman who gave birth relatively early, but later still developed
breast cancer, the number of mammary gland progenitors were again
observed to be higher than average.
“The reason we are excited about this research is that we can use a
progenitor cell census to determine who's at particularly high risk for
breast cancer,” said Polyak, a Harvard Stem Cell Institute Principal
Faculty member and Harvard Medical School professor at the Dana-Farber
Cancer Institute. “We could use this strategy to decrease cancer risk
because we know what regulates the proliferation of these cells and we
could deplete them from the breast.”
Research shows that two trends are contributing to an increase in the
number of breast cancer diagnoses - a rise in obesity and the
ever-increasing number of women postponing child bearing. The
scientists’ long-range goal is to develop a protective treatment that
would mimic the protective effects of early child bearing. The research,
which took five years to complete, began with conversations between
Polyak and John Hopkins University School of Medicine Professor
Saraswati Sukumar.
The two scientists formed collaborations with clinicians at cancer
centres that see large numbers of high-risk women in order to obtain
breast tissue samples. They also worked with genomics experts and
bioinformaticians to analyse gene expression in different breast cell
types. At times, Polyak and Sukumar had trouble convincing others to
help with the study, which is unique in the breast cancer field for its
focus on risk prediction and prevention.
“In general people who study cancer always want to focus on treating
the cancer but in reality, preventing cancer can have the biggest impact
on cancer-associated morbidity and mortality,” Polyak said. “I think the
mentality has to change because breast cancer affects so many women, and
even though many of them are not dying of breast cancer, there's a
significant personal and societal burden.”
- MNT
Obese mothers' children ‘more likely to die young’
Children born to obese mothers are 35 percent more likely to die
before they reach 55, a study has found.
They also have a 29 percent increased chance of being admitted to
hospital for heart attacks, angina and stroke than those born to mothers
of a normal weight. Experts analysed data for 37,709 babies delivered
between 1950 and 1976 in Scotland. Their mother’s weight was recorded
during her first antenatal appointment in pregnancy.
The results showed that offspring were 35 percent more likely to have
suffered an early death by the age of 55 if their mother had been obese
in pregnancy (body mass index of 30 or over). This held true even after
other factors, including mother’s age, socio-economic status, sex of the
child and current weight, were taken into account. Writing in the
British Medical Journal , the experts concluded: “Maternal obesity is
associated with an increased risk of premature death in adult
offspring.” Among the 28,540 mothers, 21 percent (5,993) were overweight
and four percent (1,141) were obese.The researchers, from the
universities of Aberdeen and Edinburgh, said the results were a “major
public health concern”, especially seeing as only four percent of
mothers in the study were obese, “far smaller than current levels in the
US and UK”.
- The Independent
|


