|

Lab-grown egg cells could
revolutionise fertility and even banish menopause :
Scientists rewrite rules of human reproduction
by Steve CONNOR
The first human egg cells that have been grown entirely in the
laboratory from stem cells could be fertilised later this year in a
development that will revolutionise fertility treatment and might even
lead to a reversal of the menopause in older women. Scientists are about
to request a licence from the UK fertility watchdog to fertilise the
eggs as part of a series of tests to generate an unlimited supply of
human eggs, a breakthrough that could help infertile women to have
babies as well as making women as fertile in later life as men.
 Producing human eggs from stem cells would also open up the
possibility of replenishing the ovaries of older women so that they do
not suffer the age-related health problems associated with the
menopause, from osteoporosis to heart disease. Producing human eggs from stem cells would also open up the
possibility of replenishing the ovaries of older women so that they do
not suffer the age-related health problems associated with the
menopause, from osteoporosis to heart disease.
Some scientists are even suggesting the possibility of producing an
“elixir of youth” for women, where the menopause is eradicated and older
women will retain the health they enjoyed when younger.
Researchers at Edinburgh University are working with a team from
Harvard Medical School in Boston to be the first in the world to produce
mature human eggs from stem cells isolated from human ovarian tissue.
Until now, it has only been possible to isolate a relatively small
number of mature human egg cells directly from the ovaries of women who
have been stimulated with hormones. This technical limitation has led to
an acute shortage of human eggs, or “oocycts”, for IVF treatment as well
as scientific research.
The scientists want to fertilise the laboratory-grown egg cells with
human sperm to prove that they are viable. Any resulting embryos will be
studied for up to 14 days - the legal limit - to see if they are normal.
These early embryos will not be transplanted into a woman's womb
because they will be deemed experimental material, but will either be
frozen or allowed to perish.
Evelyn Telfer, a reproductive biologist at Edinburgh University, has
already informally approached the Human Fertilisation and Embryology
Authority (HFEA) with a view to submitting a formal licence application
within the next few weeks.
“We hope to apply for a research licence to do the fertilisation of
the in vitro grown oocytes within the IVF unit at the Edinburgh Royal
Infirmary,” Dr Telfer said.
“Could the fertilisation take place this year? Yes, absolutely,” she
said. Professor Richard Anderson of the MRC Centre for Reproductive
Health, who will be in charge of the clinical aspects of the work, said:
“The aim will be to demonstrate that the eggs that we’ve generated in
vitro are competent to form embryos and that’s the best test that an egg
is an egg,”
Generating an unlimited supply of human eggs and the prospect of
reversing the menopause was made possible by a series of breakthroughs
led by Professor Jonathan Tilly of Harvard.
In 2004, he astounded the world of reproductive biology by suggesting
that there were active stem cells in the ovaries of mice that seemed
capable of replenishing eggs throughout life.
For half a century, a dogma of reproductive biology was that women
are born with their full complement of egg cells which they gradually
lose through life until they run out when they reach the menopause.
“This age-old belief that females are given a fixed ‘bank account’ of
eggs at birth is incorrect,” Professor Tilly said.
“In fact ovaries in adulthood are probably more closely matched to
testes in adulthood in their capacity to make new germ cells, which are
the special cells that give rise to sperm and eggs,“ he said.
”Over the past 50 years, all the basic science, all the clinical work
and all the clinical outcome was predicated on one simple belief, that
is the oocyte pool, the early egg-cell pool in the ovaries was a fixed
entity, and once those eggs were used up they cannot be renewed,
replenished or replaced,“ he added.
Last month, Prof Tilly published pioneering research showing that
these stem cells exist in human ovaries and that they could be
stimulated in the laboratory to grow into immature egg cells.
He is collaborating with Dr Telfer, who was once sceptical of his
research, because in Edinburgh she has pioneered a technique for growing
immature eggs cells to the fully “ripened” stage when they can be
fertilised.
”It's been fun to work with her because she's been one of the most
vocal critics of this work years ago and it's great that she's come
about and changed her views,“ Prof Tilly said.
”I think personally [fertilising the first eggs] is do-able. I see no
hurdles why it cannot be done this year,“ he said.
Dr Telfer added: “The important thing is that if you can show you can
get ovarian stem cells from human ovary you then have the potential to
do more for fertility preservation.
“We have all the local ethical approval in place and we’re now
looking at the process of the HFEA application. There is a push for us
to do it now,” she added.
The Independent
The mystery of human consciousness
A wakening from anesthesia is often associated with an initial phase
of delirious struggle before the full restoration of awareness and
orientation to one's surroundings. Scientists now know why this may
occur: primitive consciousness emerges first. Using brain imaging
techniques in healthy volunteers, a team of scientists led by Adjunct
Prof Harry Scheinin, M.D. from the University of Turku, Turku, Finland
in collaboration with investigators from the University of California,
Irvine, USA, have now imaged the process of returning consciousness
after general anesthesia. The emergence of consciousness was found to be
associated with activations of deep, primitive brain structures rather
than the evolutionary younger neocortex.
 These results may represent an important step forward in the
scientific explanation of human consciousness. The study was part of the
Research Program on Neuroscience by the Academy of Finland. “We expected
to see the outer bits of brain, the cerebral cortex (often thought to be
the seat of higher human consciousness), would turn back on when
consciousness was restored following anesthesia. Surprisingly, that is
not what the images showed us. In fact, the central core structures of
the more primitive brain structures including the thalamus and parts of
the limbic system appeared to become functional first, suggesting that a
foundational primitive conscious state must be restored before higher
order conscious activity can occur” Scheinin said. These results may represent an important step forward in the
scientific explanation of human consciousness. The study was part of the
Research Program on Neuroscience by the Academy of Finland. “We expected
to see the outer bits of brain, the cerebral cortex (often thought to be
the seat of higher human consciousness), would turn back on when
consciousness was restored following anesthesia. Surprisingly, that is
not what the images showed us. In fact, the central core structures of
the more primitive brain structures including the thalamus and parts of
the limbic system appeared to become functional first, suggesting that a
foundational primitive conscious state must be restored before higher
order conscious activity can occur” Scheinin said.
Twenty young healthy volunteers were put under anesthesia in a brain
scanner using either dexme-detomidine or propofol anesthetic drugs. The
subjects were then woken up while brain activity pictures were being
taken. Dexmedetomidine is used as a sedative in the intensive care unit
setting and propofol is widely used for induction and maintenance of
general anesthesia.
Dexmedetomidineinduced unconsciousness has a close resemblance to
normal physiological sleep, as it can be reversed with mild physical
stimulation or loud voices without requiring any change in the dosing of
the drug. This unique property was critical to the study design, as it
enabled the investigators to separate the brain activity changes
associated with the changing level of consciousness from the drugrelated
effects on the brain. The staterelated changes in brain activity were
imaged with positron emission tomography (PET).
The emergence of consciousness, as assessed with a motor response to
a spoken command, was associated with the activation of a core network
involving subcortical and limbic regions that became functionally
coupled with parts of frontal and inferior parietal cortices upon
awakening from dexme-detomidine-induced unconsciousness. This network
thus enabled the subjective awareness of the external world and the
capacity to behaviorally express the contents of consciousness through
voluntary responses.
Interestingly, the same deep brain structures, i.e. the brain stem,
thalamus, hypothalamus and the anterior cingulate cortex, were activated
also upon emergence from propofol anesthesia, suggesting a common,
drugindependent mechanism of arousal. For both drugs, activations seen
upon regaining consciousness were thus mostly localized in deep,
phylogenetically old brain structures rather than in the neocortex. The
researchers speculate that because current depth-of-anesthesia
monitoring technology is based on cortical electroencephalography (EEG)
measurement (i.e., measuring electrical signals on the sur-face of the
scalp that arise from the brain's cortical surface), their results help
to explain why these devices fail in differentiating the conscious and
unconscious states and why patient awareness during general anesthesia
may not always be detected. The results presented here also add to the
current understanding of anesthesia mechanisms and form the foundation
for developing more reliable depth-of-anesthesia technology.
The anesthetised brain provides new views into the emergence of
consciousness. Anesthetic agents are clinically useful for their
remarkable property of being able to manipulate the state of
consciousness. When given a sufficient dose of an anesthetic, a person
will lose the precious but mysterious capacity of being aware of one's
own self and the surrounding world, and will sink into a state of
oblivion. Conversely, when the dose is lightened or wears off, the brain
almost magically recreates a subjective sense of being as experience and
awareness returns. The ultimate nature of consciousness remains a
mystery, but anesthesia offers a unique window for imaging internal
brain activity when the subjective phenomenon of consciousness first
vanishes and then re-emerges. This study was designed to give the
clearest picture so far of the internal brain processes involved in this
phenomenon.
The results may also have broader implications. The demonstration of
which brain mechanisms are involved in the emergence of the conscious
state is an important step forward in the scientific explanation of
consciousness. Yet, much harder questions remain. How and why do these
neural mechanisms create the subjective feeling of being, the awareness
of self and environment the state of being conscious?
NYT
A link between atherosclerosis and autoimmunity
Those who suffer from autoimmune diseases also display a tendency to
develop atherosclerosis - the condition popularly known as hardening of
the arteries. Clinical researchers at LMU, in collaboration with
colleagues in Würzburg, have now discovered a mechanism which helps to
explain the connection between the two types of disorder.
 The link is provided by a specific class of immune cells called
plasmacytoid dendritic cells (pDCs). pDCs respond to DNA released from
damaged and dying cells by secreting interferon proteins which stimulate
the immune reactions that underlie autoimmune diseases. The new study
shows that stimulation of pDCs by a specific DNA-protein complex
contributes to the progression of atherosclerosis. The link is provided by a specific class of immune cells called
plasmacytoid dendritic cells (pDCs). pDCs respond to DNA released from
damaged and dying cells by secreting interferon proteins which stimulate
the immune reactions that underlie autoimmune diseases. The new study
shows that stimulation of pDCs by a specific DNA-protein complex
contributes to the progression of atherosclerosis.
Implications
The findings may have implications for new strategies for the
treatment of a whole spectrum of conditions that are associated with
chronic inflammatory reactions. Atherosclerosis is a major cause of
death in Western societies. The illness is due to the formation of
insoluble deposits called atherosclerotic plaques on the walls of major
arteries as a consequence of chronic, localised inflammation reactions.
By reducing blood flow, the plaques can provoke heart attacks and
strokes. A class of immune cells called dendritic cells plays a crucial
role in facilitating the development of these plaques. The term refers
to a heterogeneous cell population that makes up part of the immune
system. Among the cell types represented in this population are the
so-called plasmacytoid dendritic cells (pDC), but their potential
significance for atherosclerosis had not been explored until now.
Disorders
A group of researchers led by Dr. Yvonne Döring in Prof Christian
Weber's department at LMU, together with a team supervised by
Privatdozentin Dr. Alma Zernecke of Würzburg University, has now shown
how pDCs promote the development of atherosclerosis – and explained why
patients with autoimmune disorders, such as psoriasis or systemic lupus
(SLE), show a predisposition to atherosclerosis.
Using laboratory mice as an experimental model, the researchers were
able to show that pDCs contribute to early steps in the formation of
athersclerotic lesions in the blood vessels. Stimulation of pDCs causes
them to secrete large amounts of interferons, proteins that strongly
stimulate inflammatory processes. The protein that induces the release
of interferons is produced by immune cells that accumulate specifically
at sites of inflammation, and mice that are unable to produce this
protein also have fewer plaques. Stimulation of pDCs in turn leads to an
increase in the numbers of macrophages present in plaques. Macrophages
normally act as a clean-up crew, removing cell debris and fatty deposits
by ingesting and degrading them.
However, they can also “overindulge”, taking up more fat than they
can digest. When this happens, they turn into so-called foam cells that
promote rather than combat atherosclerosis. In addition, activated,
mature pDCs can initiate an immune response against certain molecules
found in atherosclerotic lesions, which further exacerbates the whole
process.
Disorders
The disorders of pDCs provides the link between atherosclerosis and
autoimmune diseases. “The pDCs themselves are stimulated by the
self-antigens that set off the autoimmune reactions which result in
conditions like psoriasis and SLE,” says Döring. Indeed, it is well
known that the secretion of interferons by activated pDCs contributes to
the genesis of a number of autoimmune diseases
“The findings also suggest new approaches to the treatment of chronic
inflammation that could be useful for a whole range of diseases,” said
Webber.
MNT
Enzyme in saliva helps regulate blood glucose
Scientists from the Monell Center report that blood glucose levels
following starch ingestion are influenced by genetically determined
differences in salivary amylase, an enzyme that breaks down dietary
starches. Specifically, higher salivary amylase activity is related to
lower blood glucose.
The findings are the first to demonstrate a significant metabolic
role for salivary amylase in starch digestion, suggesting that this oral
enzyme may contribute significantly to overall metabolic status. Other
implications relate to calculating the glycemic index of starch-rich
foods and ultimately the risk of developing diabetes “Two individuals
may have very different glycemic responses to the same starchy food,
depending on their amylase levels,” said lead author Abigail Mandel,
Ph.D., a nutritional scientist at Monell.
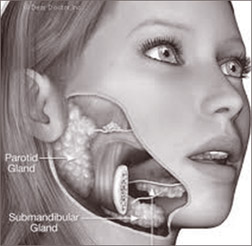 “Those with high amylase levels are better adapted to eat starches,
as they rapidly digest the starch while maintaining balanced blood
glucose levels. The opposite is true for those with low amylase levels. “Those with high amylase levels are better adapted to eat starches,
as they rapidly digest the starch while maintaining balanced blood
glucose levels. The opposite is true for those with low amylase levels.
As such, people may want to take their amylase levels into account if
they are paying attention to the glycemic index of the foods they are
eating.” Starch from wheat, potatoes, corn, rice, and other grains is a
major component of the United States diet, comprising up to 60 percent
of our calories. Amylase enzymes secreted in saliva help break down
starches into simpler sugar molecules that can be absorbed into the
bloodstream. In this way, amylase activity influences blood glucose
levels, which need to be maintained within an optimal range for good
health.
Starch
A previous study had demonstrated that individuals with high salivary
amylase activity are able to break down oral starch very rapidly. This
finding led the researchers to ask how this ‘pre-digestion’ contributes
to overall starch digestion and glucose metabolism.
In the current study, published online in *The Journal of Nutrition*,
amylase activity was measured in saliva samples obtained from 48 healthy
adults. Based on extremes of salivary amylase activity, two groups of
seven were formed: high amylase (HA) and low amylase (LA). Each subject
drank a simplified corn starch solution and blood samples were obtained
over a two hour period afterwards. The samples were analysed to
determine blood glucose levels and insulin concentrations.
After ingesting the starch, individuals in the HA group had lower
blood glucose levels relative to those in the LA group. This appears to
be related to an early release of insulin by the HA individuals.
“Not all people are the same in their ability to handle starch,” said
senior author Paul Breslin, Ph.D., a sensory geneticist at Monell.
“People with higher levels of salivary amylase are able to maintain
more stable blood glucose levels when consuming starch.
This might ultimately lessen their risk for insulin resistance and
non-insulin dependent diabetes.”
Additional studies will confirm the current findings using more
complex starchy foods, such as bread and pasta. Another focus will
involve identifying the neuroendocrine mechanisms that connect starch
breakdown in the mouth with insulin release.
- Medicalxpress
|

