Alzheimer's: New drug clears toxic proteins from patients' brains
'Compared to other studies published in the past, the effect size of
this drug is unprecedented'
An antibody that can almost completely clear the visible signs of
Alzheimer's disease from the brain has been discovered in a breakthrough
that left one scientist 'trying not to get too excited'.
Researchers scanned the brains of people with the degenerative
condition as they were given doses of the drug, which is based on an
immune cell taken from the blood of elderly people aged up to 100 who
showed no signs of the disease.
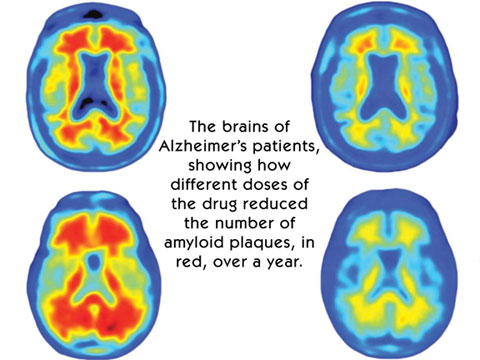 After a year, virtually all the toxic 'amyloid plaques' that build up
in Alzheimer's patients appeared to have gone from the brains of those
given the highest doses of the antibody. After a year, virtually all the toxic 'amyloid plaques' that build up
in Alzheimer's patients appeared to have gone from the brains of those
given the highest doses of the antibody.
Trials
The scientists, who described their results in a paper in Nature,
also said the patients showed signs that the rate of their cognitive
decline had slowed.
The findings of the trial suggest the plaques are at least part of
the cause of the disease - not simply a byproduct.
And, if the results are confirmed in larger clinical trials already
under way around the world, one expert said it could be a 'game-changer'
for efforts to prevent Alzheimer's.
One of the researchers, Prof. Roger Nitsch of Zurich University,
described what they found when they scanned the brains of patients given
either a placebo or three different doses of the antibody, called
aducanumab.
"One year later, the images of the placebo group are basically
unchanged. In the three doses groups, a very clear reduction in amyloid
plaques is shown - the higher the dose, the larger the degree of
reduction," he said.
Treatment
"In the 10mg dose group, after one year you can see no red on the
image, meaning the amyloid has almost completely disappeared. "Compared
to other studies published in the past, the effect size of this drug is
unprecedented." Commenting on the research in a separate Nature article,
Prof Eric Reiman, of The University of Arizona, wrote: "If these
preliminary cognitive findings are confirmed in larger and
more-definitive clinical trials, which are now under way, it would
provide a shot in the arm in the fight against Alzheimer's disease. "But
although the authors' additional cognitive findings are encouraging,
they are not definitive. It would be prudent to withhold judgement about
aducanumab's cognitive benefit until results from the larger trials are
in."
But he added: "Confirmation that an anti-amyloid plaque treatment
slows cognitive decline would be a game-changer for how we understand,
treat and prevent Alzheimer's disease.
"Now is the time to find out."
The study, led by scientists at pharmaceutical companies Biogen and
Neurimmune, sparked major interest from experts in the field.
Interfere
Dr Tara Spires-Jones, interim director of Edinburgh University's
Centre for Cognitive and Neural Systems, said the research showed the
antibody "robustly reduced amyloid pathology in a small group of people
in very early stages of the disease".
"I am cautiously optimistic about this treatment, but trying not to
get too excited because many drugs make it through this early stage of
testing then go on to fail in larger trials," she said.
And Dr James Pickett, head of research at the Alzheimer's Society,
said: "These results are the most detailed and promising that we've seen
for a drug that aims to modify the underlying causes of Alzheimer's
disease. "The study showed that the drug was first able to remove clumps
of amyloid - a toxic protein associated with Alzheimer's - from the
brain of mice and also, excitingly, in people.
"What is most compelling is that more amyloid was cleared when people
took higher doses of the drug.
"No existing treatments for Alzheimer's directly interfere with the
disease process - and so a drug that actually slows the progress of the
disease by clearing amyloid would be a significant step."
He noted that the researchers had found some side-effects, such as
headaches, and that the initial trial had not been designed to measure
whether the drug slowed the decline in memory and thinking.
"While there were hints that it might have an effect on the symptoms
of the disease, we need to see the results from further, larger research
trials to understand whether this is the case. These larger trials are
now under way, including in the UK, and due to finish in 2020," Dr
Pickett said.
However, Gordon Wilcock, emeritus professor of geratology at Oxford
University, was somewhat downbeat about the findings.
"This is preliminary data about another monoclonal antibody targeting
amyloid in Alzheimer's disease, but whether it will really produce
meaningful clinical benefit only definitive phase three clinical trials
will show, as the researchers themselves state," he said.
"We have already had previous trials of various anti-amyloid
strategies, especially the monoclonal antibodies that have failed to
deliver at phase three.
"Nevertheless these trials are justified by the data and I hope they
are successful, despite my feelings of déjà vu!"
- The Independent
|

